Draft Guidance Document: Applications for Medical Device Investigational Testing Authorizations - Canada.ca
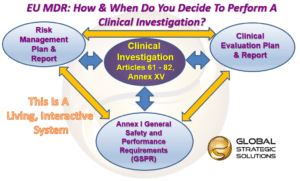
What Is The Difference Between Clinical Evaluation and Clinical Investigation? | Global Strategic Solutions

Importance of systematic literature search for clinical evaluation(ce) the strict adherence of medde by PepGra CRO - Issuu
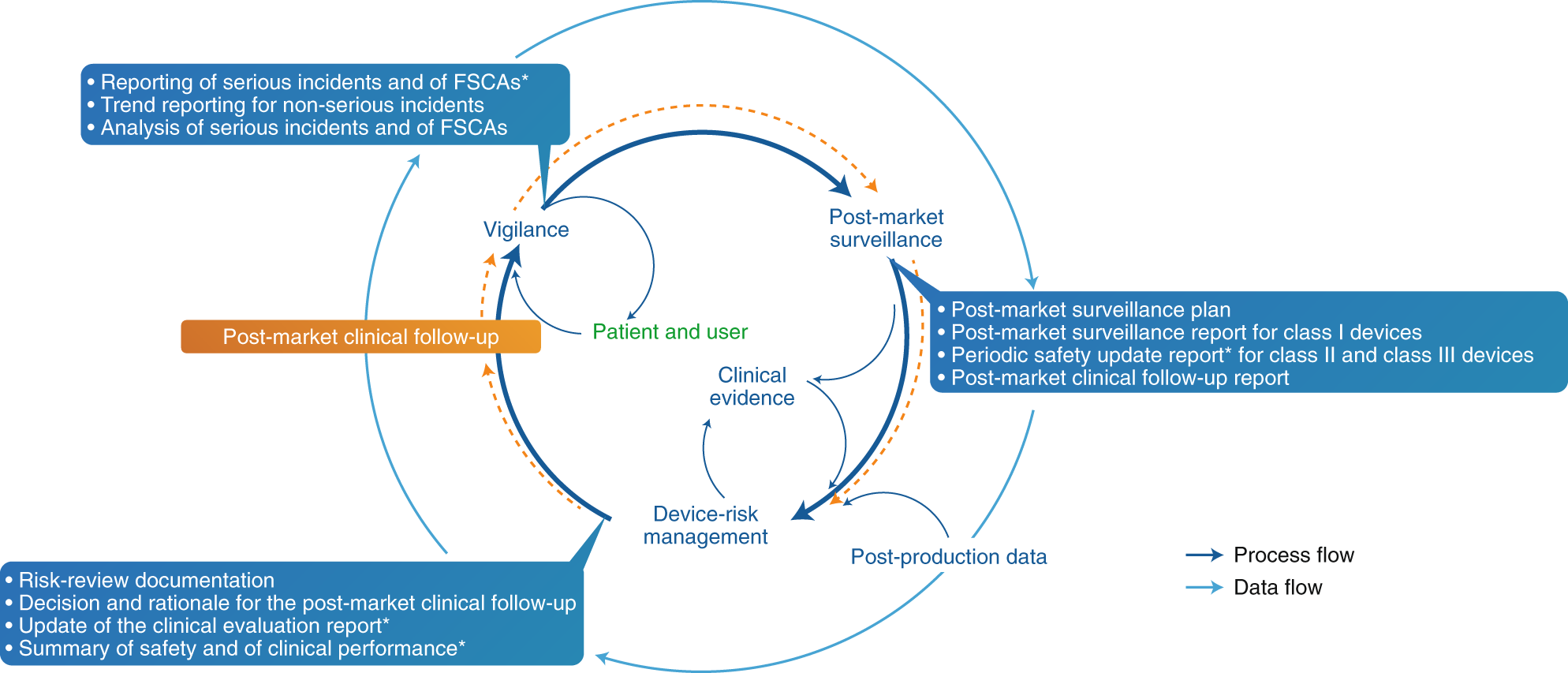
How the new European regulation on medical devices will affect innovation | Nature Biomedical Engineering

ANSI/AAMI/ISO 14155-2:2003 - Clinical investigation of medical devices for human subjects - Part 2: Clinical investigation plans

Applications for Medical Device Investigational Testing Authorizations Guidance Document - Canada.ca

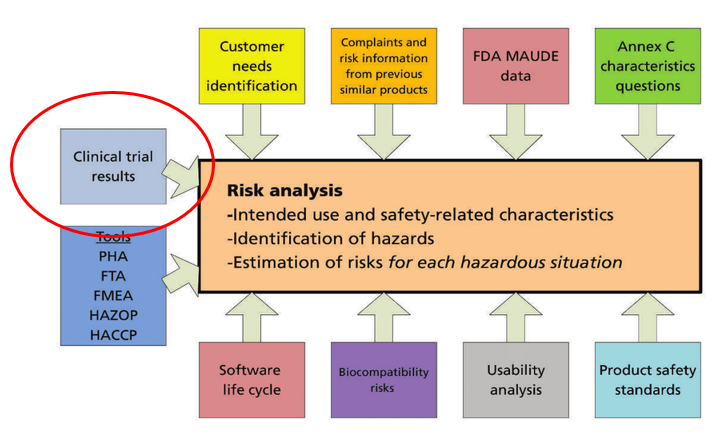
Medical Device Quality, Regulatory and Product Development Blog | Greenlight Guru | QMS Software (4)